Dec 24 2015
2 x 30 = $6.48
Tuesday
No training. I had a 3x20min on the program but decided to skip it. The body felt too tired and this is a light week so no big harm done.
Wednesday
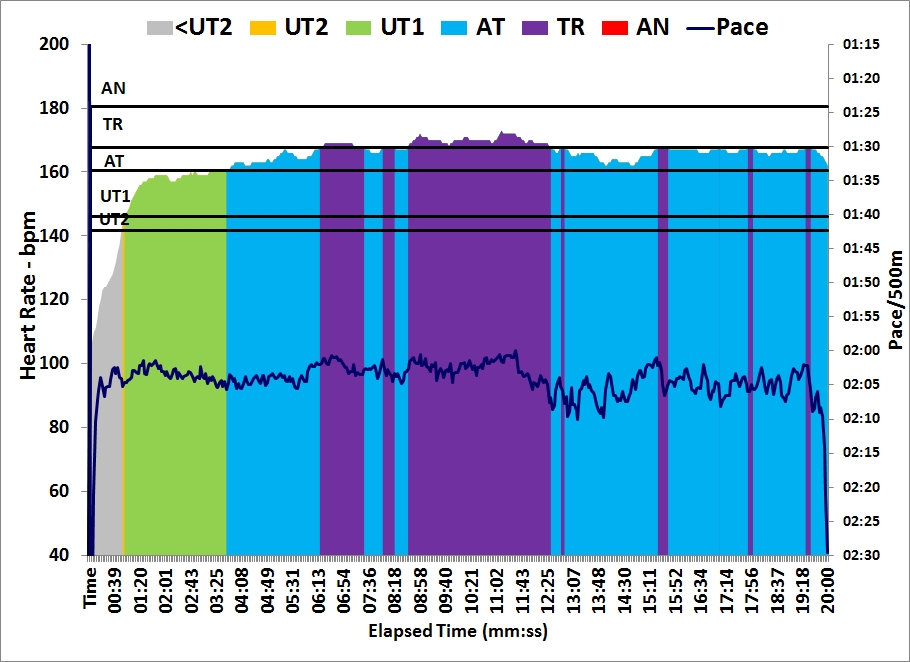
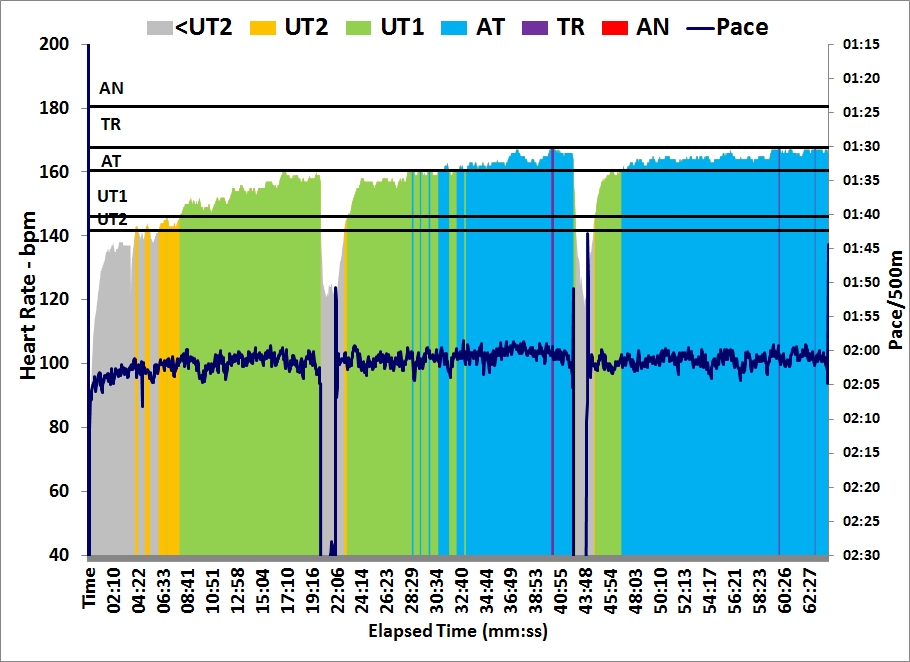
Last day at work before the Christmas break. At home I did a 30 minute
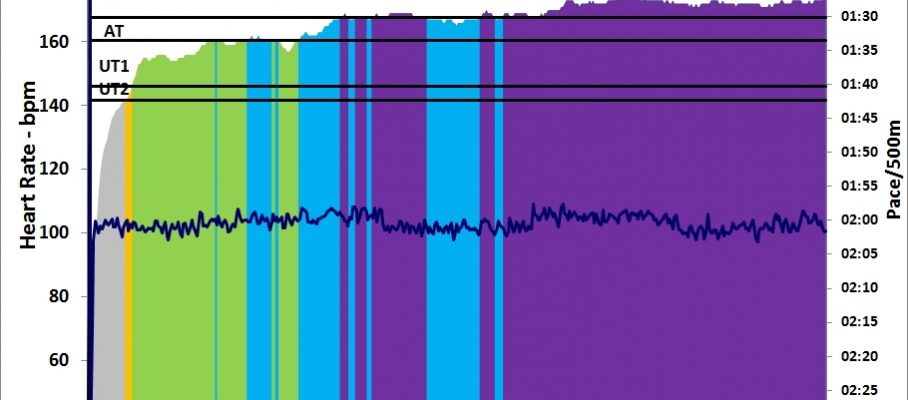
row followed by a 30 minute strength training. I used the “heavier” bands and fewer repetitions.
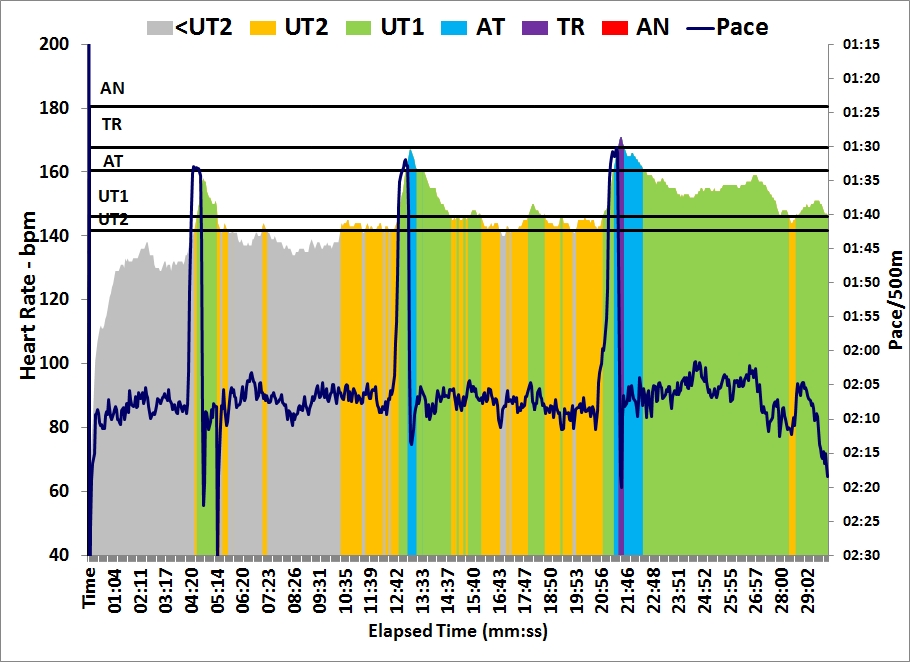
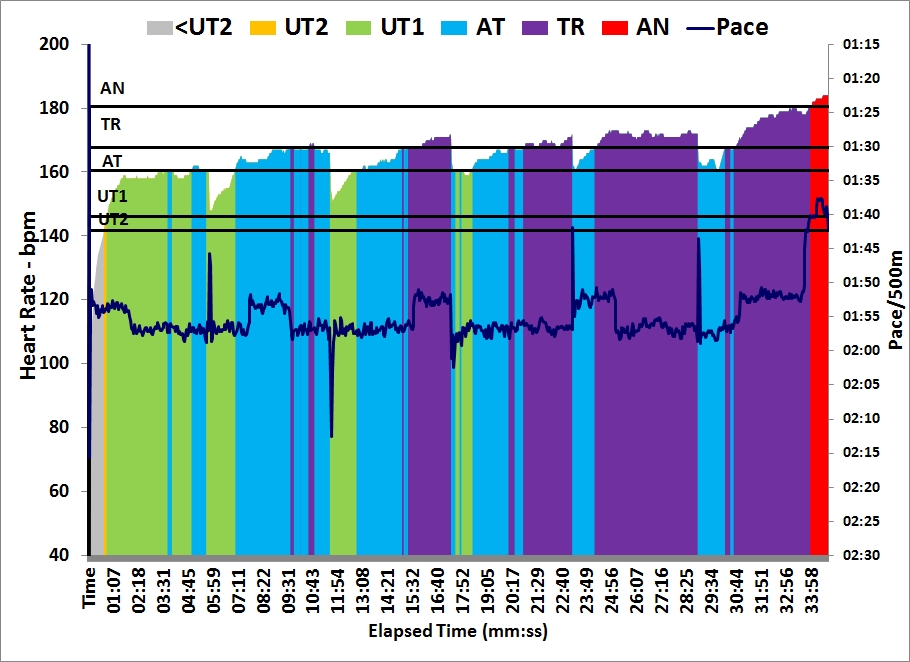
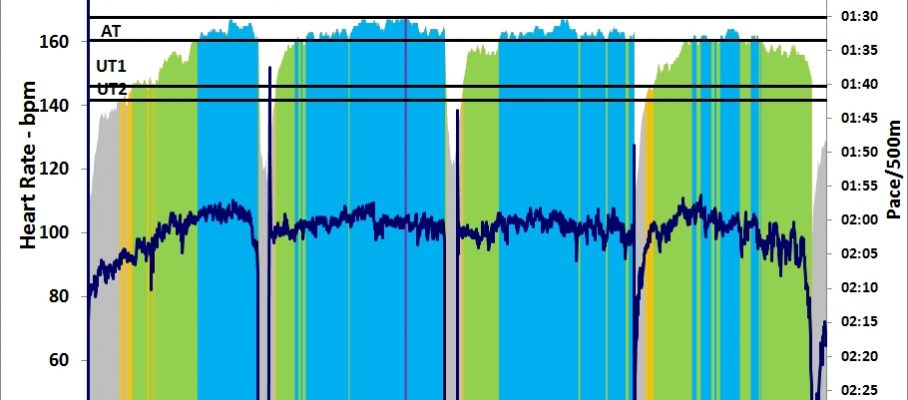
As you can see from the graph, I spiced up my 30 minutes of light rowing with a few 10 stroke sprints:
Thursday (Christmas Eve)
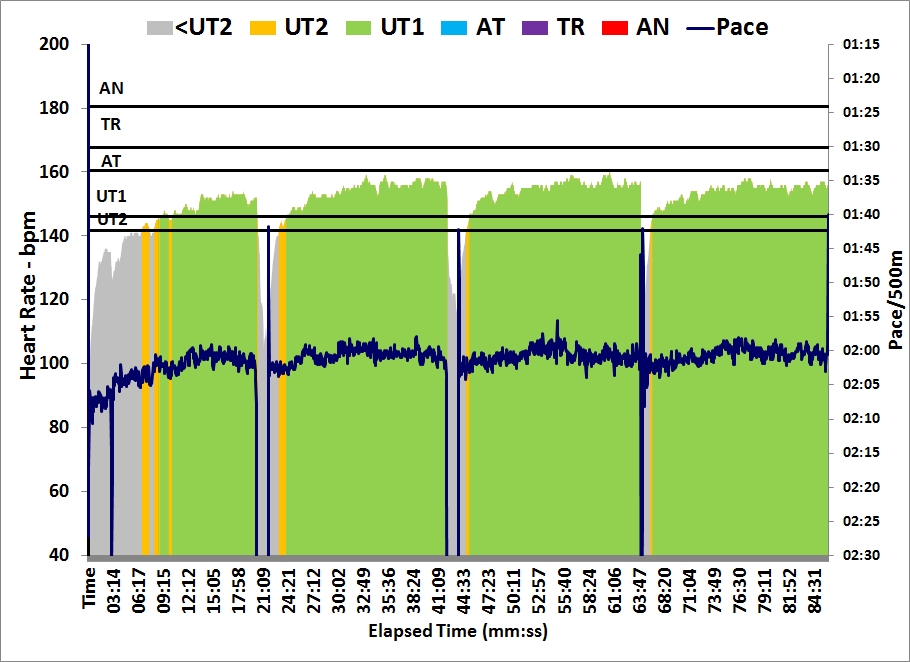
In the morning, Romana and I went to a Christmas event at the Lodni Sporty rowing club. We parked the car a good distance from the club and walked along the lake. There was thick fog, visibility about 25 meters, and the water was mirror flat.
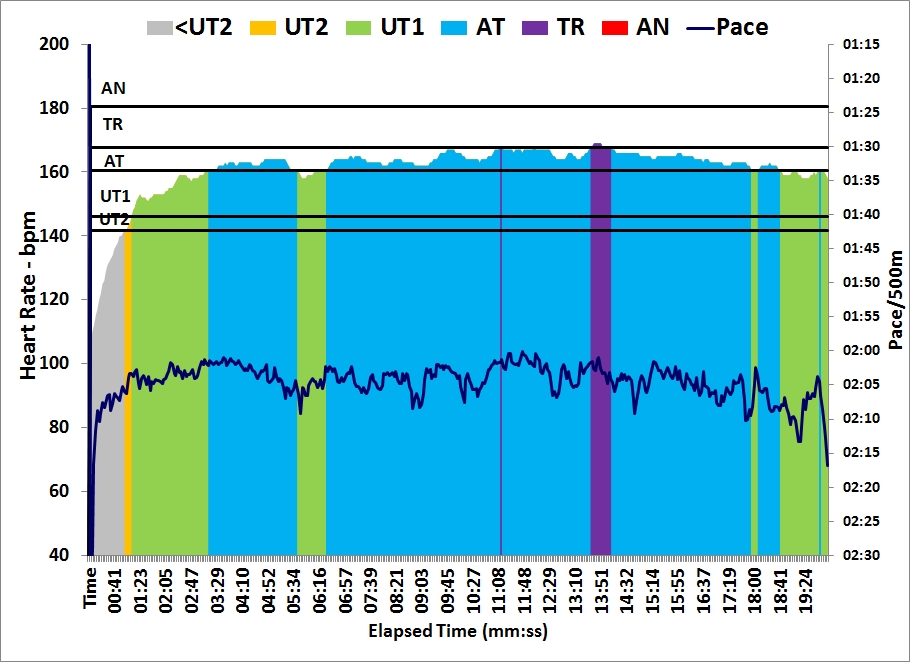
In the afternoon I did a 30 minutes easy row. I did a few 500s at slightly faster pace, to catch up with my pace boat.
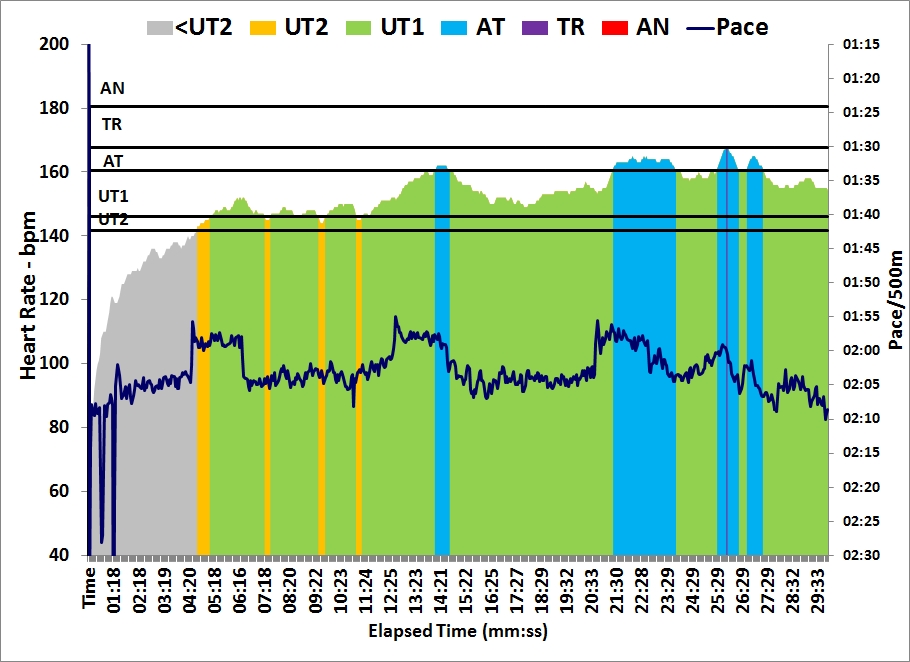
These two rows brought me past the 200 km mark for the Concept2 holiday challenge. I have now rowed $6.48 in money raised by Concept2.
Considering my heavy travel program over the past month, I consider this a success.
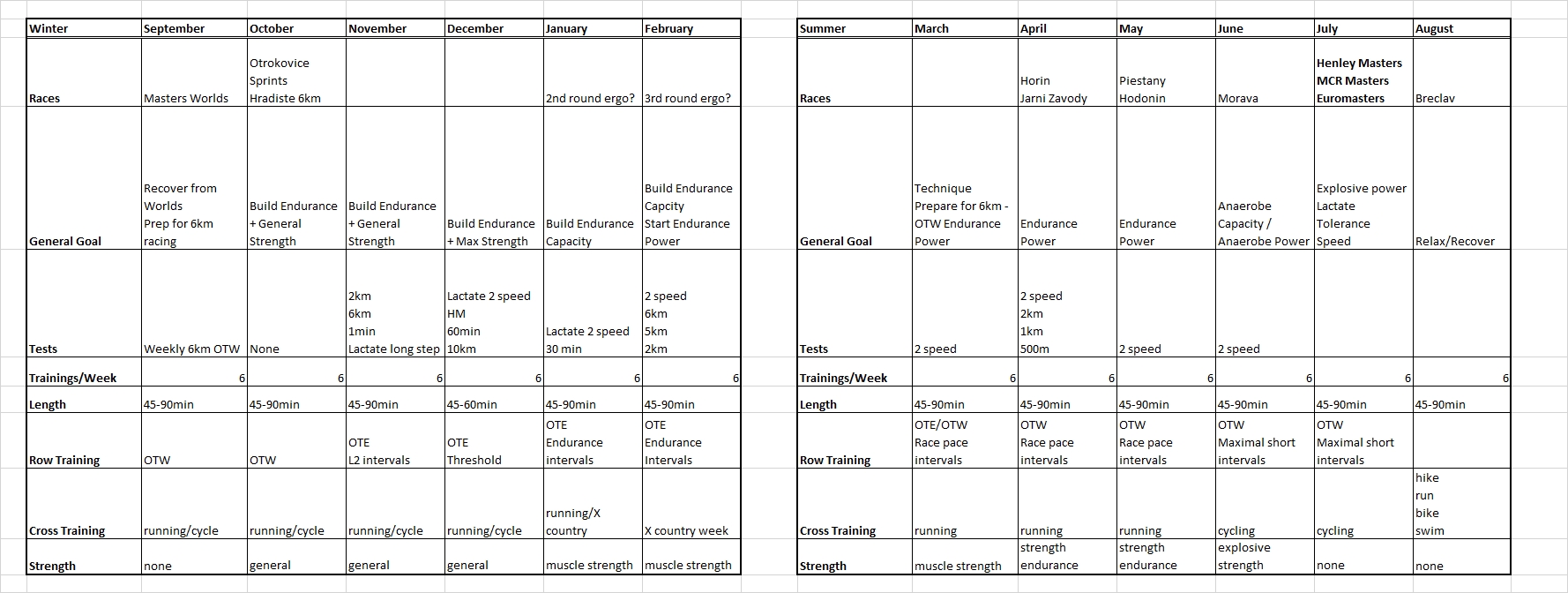
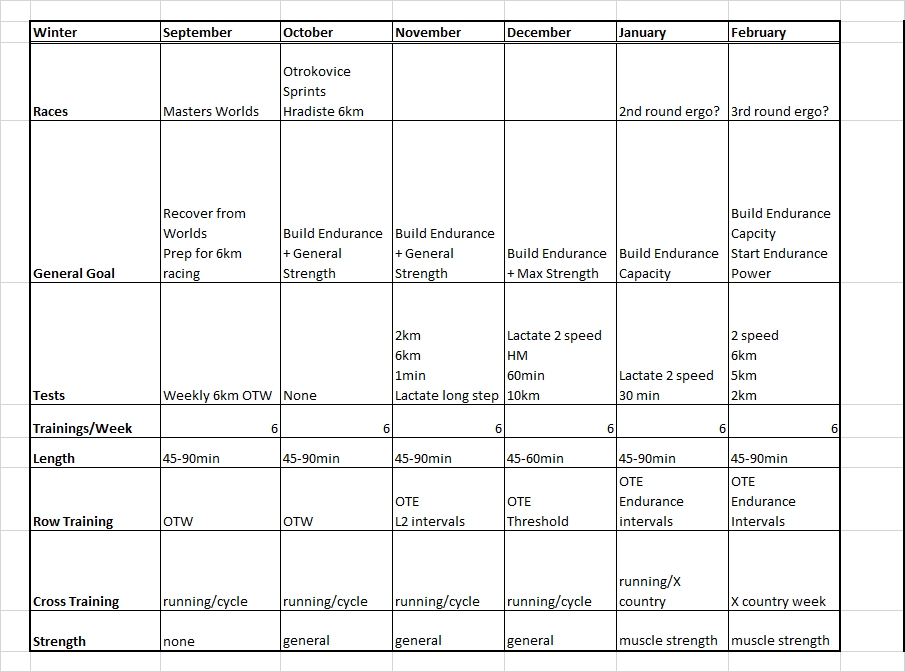
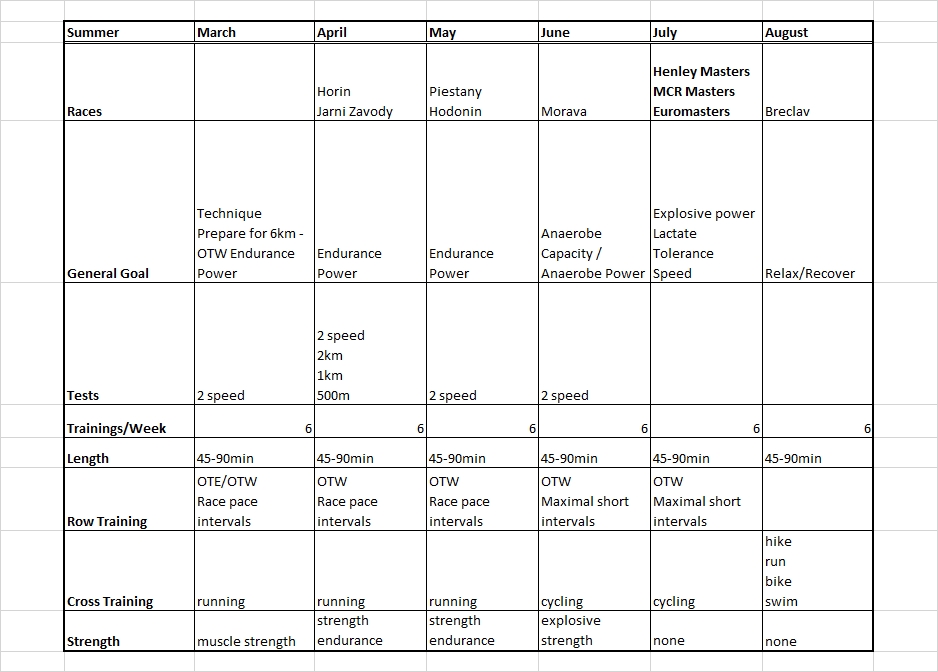




Dec 29 2015
4x20min at lactate
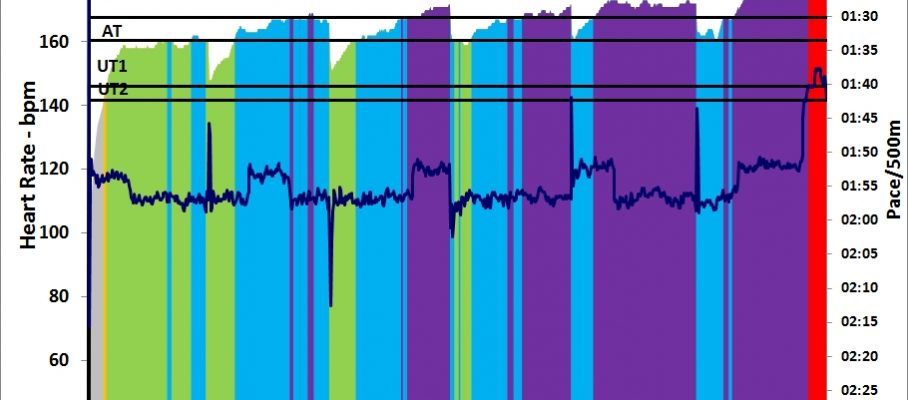
Managed to get RowPro not crashing, so I did a 1k warming up and then the main thing.
RowPro behaved well until it wanted to save stroke data for the row. It threw an error message and offered that I email this to assist at digitalrowing.com, which I did. In the rowing log it shows up as 0m rowed, but when I export a CSV file, I have the summary data, at least:
I started slowly, increasing from 170W to 200W during the first interval. Then I remained between 195W and 205W in intervals #2 and #3. This did feel a bit harder than it should and my heart rate seemed high. Did a Lactate measurement and got my suspicion confirmed: 1.7 mmol/L is a high value for me. I want to be around 1.2-1.5 mmol/L.
So I gave myself permission to back down during the final interval, which I rowed without much pace discipline, oscillating between 170W and 200W. Measured lactate again with the last strip from my first batch and got a nice value of 0.9 mmol/L.
I had some difficulties measuring lactate in the past few weeks. It has been very humid weather. The meter gave an error message indicating that it thought the strips were already used. I suspect that lying for an hour in my humid basement didn’t do them good. So I threw those strips away. Yesterday, I replaced the thermometer with a full blown weather station including a measurement of humidity. When I started today’s row, the air in my basement had a relative humidity of 61% at a temperature of 9 degrees C. This corresponded nicely with the Brno airport reported dew point of 2 degrees C (and an outside temperature of 4 degrees, and thus a partial H2O pressure of 7 hPa). During the row, the temperature rose by 0.1 degree but the relative humidity increased to 70%, I guess by my own breathing. In these conditions, the strips turned out to be usable, but I suspect that in the past weeks the relative humidity has been well above 85%, which is the maximum according to the spec of the strips.
Now I have RowPro misbehaving again. I may try to delete the database and see if that makes RowPro behave well again. Alternatively, I may stop using RowPro and just use the PM.
The training program says that I need to test a 60 minute row this week, but that was just to get nonathlon points. I looked at my half marathon result and decided that I would put a pro-rated score of 15319m on nonathlon. I also noticed that during the 60 minutes of my half marathon I covered 15380m, which is 37m past my PB. So I consider that a new Personal Best.
So I dropped the 60min. I moved a 5k test to January because it is that month’s CTC. I also moved the 10k to the next mesocycle where it will be a nice threshold training. That improves the focus on anaerobic capacity for this mesocycle, which is good.
By sanderroosendaal • Uncategorized • 0 • Tags: concept2, erg, lactate, OTE, rowing, steady state